-
Kai Trump faces backlash over luxury Erewhon video - 20 mins ago
-
Dabble Promo Code FOX: $10 No Deposit Bonus for New Users - 23 mins ago
-
More than 70 dead amid massive winter storm as freezing temperatures remain - 25 mins ago
-
Rihanna’s home targeted in Beverly Hills shooting, suspect charged - 28 mins ago
-
Wrong kind of fuel: Nearly 1,000 pounds of liquid meth seized in big rig fuel tank at Otay Mesa - 57 mins ago
-
Star Team Paris Saint-Germain and Spartak Moscow to Play in Youth Tournament in Budapest - about 1 hour ago
-
2026 NFL Odds: Did Bills Truly Land New No. 1 Wideout? - about 1 hour ago
-
John Tesh survives decade after 18-month cancer prognosis in 2015 - about 1 hour ago
-
Another major winter storm, with possible blizzard conditions, pushes into the Southeast - about 1 hour ago
-
Anthropic sues Pentagon, Trump administration over “supply chain risk” designation - about 1 hour ago
Diamonds Are Even More Forever as Scientists Create Special Harder Variety
ÔÇťWhen loveÔÇÖs gone, they luster on,ÔÇŁ sang Shirley┬áBassey in 1971ÔÇÖs “Diamonds Are Forever.” Well, come 2026, that maxim might be truerÔÇöwith researchers in China reporting that they have succeeded in artificially creating a special, harder variety of the carbon-based gem.
Long regarded as the hardest mineral, the diamonds that we are used to have a cubic crystal structure. There is, however, a rare and potentially tougher form with a hexagonal structure.
Also known as lonsdaleite, hexagonal diamond is found in nature when meteorites made up of (carbon-based) graphite slam into earth and experience extreme temperatures and pressures. 
Lonsdaleite has been hard to study, however, because it is produced this way in such small quantitiesÔÇöleading some to suggest that samples of hexagonal diamond are in fact just cubic diamond with internal stacking faults or nanoscale twinning.
In a new study, however, researchers from China believe they have settled the debate for once and for all by manufacturing bulk sample of hexagonal diamond in the lab.
ÔÇťThese findings resolve the long-standing controversy on the existence of hexagonal diamond as a discrete carbon phase,ÔÇŁ the researchers wrote in their paper.
In addition, they continued, the work provides ÔÇťnew insights into the graphite-to-diamond phase transition, paving the way for future research and practical use of hexagonal diamond in advanced technological applications.ÔÇŁ
Such applications for hexagonal diamond could include in cutting and drilling tools, within high-end electronics, and for the production of specialist industrial and scientific equipment.
To make the rare material in the laboratory, material scientist Shoulong Lai of ChinaÔÇÖs Zhengzhou University and colleagues started with a different form of carbonÔÇöÔÇŁhighly oriented pyrolytic graphite,ÔÇŁ which is not dissimilar to the graphite found in pencils.
This highly ordered graphite was placed between two anvils made of the extremely hard material tungsten carbide and subjected to pressures of 20 gigapascalsÔÇöthat is, nearly 200,000 times atmospheric pressureÔÇöand temperatures between 1,292ÔÇô3,452 degrees Fahrenheit.
The graphite was placed within the anvil such that the pressure was applied to the top and bottom of the stacked carbon layers making up the material, rather than the sides.
In this way, the researchers say they have managed to produce millimeter-sized samples of pure hexagonal diamondÔÇöan outcome they report confirming by probing the materialÔÇÖs structure using a combination of X-ray diffraction and electron microscopy.
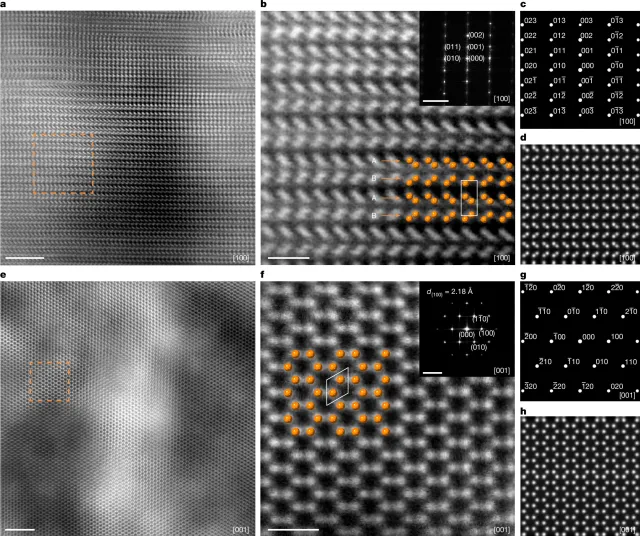
The researchers also assessed the hardness of their hexagonal diamond samples using the so-called Vickers hardness test.
This assessmentÔÇödeveloped by researchers at the British engineering firm Vickers Ltd. in the early 1920sÔÇöinvolved poking a sample with a small diamond, and measuring how much of an indent is left behind.
According to the team, their hexagonal diamond samples have a hardness of around 114 gigapascals.
This is slightly harder than natural, cubic, diamond, which typically have a hardness of 110 gigapascals.
ÔÇťHexagonal diamond exhibits a hardness value slightly exceeding that of cubic, together with a high shear modulus and notable thermal stability,ÔÇŁ the researchers wrote.
Do you have a tip on a science story that Newsweek should be covering? Do you have a question about mineralogy? Let us know via science@newsweek.com.
Reference
Lai, S., Yang, X., Shi, J., Liu, S., Guo, Y., Yan, L., Zang, J., Zhang, Z., Jia, Q., Sun, J., Cheng, S., & Shan, C. (2026). Bulk hexagonal diamond. Nature. https://doi.org/10.1038/s41586-026-10212-4
Source link































